Transient Opening of the Mitochondrial Permeability Transition Pore Induces Microdomain Calcium Transients in Astrocyte Processes.
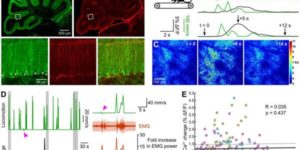
Astrocytes extend highly branched processes that form functionally isolated microdomains, facilitating local homeostasis by redistributing ions, removing neurotransmitters, and releasing factors to influence blood flow and neuronal activity. Microdomains exhibit spontaneous increases in calcium (Ca2+), but the mechanisms and functional significance of this localized signaling are unknown. By developing conditional, membrane-anchored GCaMP3 mice, we found that microdomain activity that occurs in the absence of inositol triphosphate (IP3)-dependent release from endoplasmic reticulum arises through Ca2+ efflux from mitochondria during brief openings of the mitochondrial permeability transition pore. These microdomain Ca2+ transients were facilitated by the production of reactive oxygen species during oxidative phosphorylation and were enhanced by expression of a mutant form of superoxide dismutase 1 (SOD1 G93A) that causes astrocyte dysfunction and neurodegeneration in amyotrophic lateral sclerosis (ALS). By localizing mitochondria to microdomains, astrocytes ensure local metabolic support for energetically demanding processes and enable coupling between metabolic demand and Ca2+ signaling events.